jnmaciuch
Senior Member (Voting Rights)
[Abstract] Sera from dermatomyositis patients induce muscle weakness via activation of type I interferon (IFN) receptors
Abstract/poster from the American College of Rheumatology 2025 conferenceSuchada Kaewin, Cecilia Leijding, Kristofer Andreasson, Helene Alexanderson, Stefano Gastaldello, Ingrid Lundberg and Daniel C. Andersson
Background/Purpose: Dermatomyositis (DM) is a major subtype of idiopathic inflammatory myopathies (IIMs) and characterized by muscle weakness, systemic inflammation and cutaneous lesions. Expression of type I interferon (IFN)-stimulated genes have been found in muscle, skin tissue and peripheral blood cells which links to disease activity in DM patients. However, the mechanisms of muscle weakness in DM are incompletely understood. For instance, it is unclear to what degree systemic factors, eg IFN signalling, contribute to muscle weakness and disease phenotypes. This study aims to investigate whether inhibition of type I IFN signaling protects DM serum-induced muscle weakness and to determine the effects of IFN-a and/or IFN-b on muscle force generation.
Methods: An experimental platform was used in which isolated mouse muscles were exposed to DM patient serum or healthy control serum. Flexor digitorum brevis (FDB) muscles were isolated from healthy C57BL/6JRj mice and exposed to 10% dilution of serum from healthy controls or serum from DM patients for 24 hours. To determine whether inhibition of type I IFN protect DM serum-induced muscle weakness, an IFN-a/b receptor 1 (IFNR) blocking antibody was used. Isolated FDB muscles were pre-incubated with IFNAR1 antibody for 2 h and thereafter muscles were incubated in serum from DM patients with or without IFNAR1 antibody. To investigate the effect of IFN-a and/or IFN-b on muscle force generation, FDB muscles were incubated in 10% healthy control serum containing of 1,000U/ml of IFN-a and/or IFN-b. Muscle force was measured before and after incubation in a paired fashion.
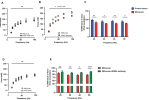
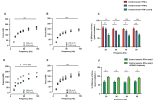
Results: Serum from DM patients, but not from healthy controls, induced a decline in FDB muscle force. Treatment with an anti-IFN1R antibody protected from the DM serum-induced muscle force decline. Surprisingly, neither IFN-a nor IFN-b affected force production when incubated for 24h in isolation with the muscles. However in combination, IFN-a together with IFN-b, induceed a marked reduction in muscle force, which was rescued with addition of an IFN1R blocking antibody.
Conclusion: Serum from DM patients contain factors that can induce muscle weakness. One mechanism for this effect is mediated by pathways dependent on type I IFN Receptor signaling.
Link