Mij
Senior Member (Voting Rights)
There are no FDA-approved treatments yet for the disease known for affecting Celine Dion
A living drug could dial back the debilitating symptoms of stiff person syndrome.
After just a single infusion, patients with the autoimmune disorder saw meaningful improvements in mobility, stiffness and disability, neurologist Amanda Piquet reported April 21 at the annual American Academy of Neurology meeting in Chicago.
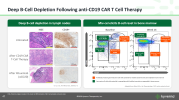
The drug, an experimental cell therapy called miv-cel, stands out from current treatment options for several reasons — including how well it appears to work. “The results of this trial are truly remarkable,” Piquet, of the University of Colorado Anschutz in Aurora, said at an April 13 news conference. “The magnitude and consistency of functional improvement observed is unprecedented.”
"Kyverna plans to request FDA approval for miv-cel in the first half of 2026. If approved, the therapy would be the first CAR T cell therapy for autoimmune disease."
A living drug could dial back the debilitating symptoms of stiff person syndrome.
After just a single infusion, patients with the autoimmune disorder saw meaningful improvements in mobility, stiffness and disability, neurologist Amanda Piquet reported April 21 at the annual American Academy of Neurology meeting in Chicago.
The drug, an experimental cell therapy called miv-cel, stands out from current treatment options for several reasons — including how well it appears to work. “The results of this trial are truly remarkable,” Piquet, of the University of Colorado Anschutz in Aurora, said at an April 13 news conference. “The magnitude and consistency of functional improvement observed is unprecedented.”
"Kyverna plans to request FDA approval for miv-cel in the first half of 2026. If approved, the therapy would be the first CAR T cell therapy for autoimmune disease."