Abstract
Chimeric antigen receptor (CAR)-T cells are considered a powerful therapeutic tool to reset the immune system in patients with autoimmune diseases. Innovative trial designs are needed to allow feasible testing of the safety and efficacy of CAR-T cells in clinical studies. CASTLE (CAR-T cells in systemic B cell mediated autoimmune disease) is a phase 1/2a two-stage optimal design basket study that investigated the safety and efficacy of zorpocabtagene autoleucel (Zorpo-cel, also known as MB-CART19.1), an autologous CD19 CAR-T cell product, in patients with treatment-resistant systemic lupus erythematosus (SLE), systemic sclerosis (SSc) and idiopathic inflammatory myopathies (IIM). The primary safety outcome was the rate of cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS). The secondary clinical efficacy outcomes were remission of SLE according to DORIS criteria, no progression of interstitial lung disease in SSc and American College of Rheumatology (ACR) major/moderate response in IIM after 24 weeks. A total of 24 patients were enrolled (10 with SLE, 9 with SSc and 5 with IIM), all receiving a single infusion of Zorpo-cel after stopping immunosuppressive treatments and receiving standard lymphodepletion with cyclophosphamide and fludarabine. Primary and secondary endpoints of CASTLE were met. Regarding safety, no CRS higher than grade 2 and no ICANS occurred. Regarding efficacy, 22 of the 24 patients achieved predefined efficacy endpoints, with 9 out of 10 patients with SLE reaching DORIS remission, 9 out of 9 patients with SSc showing no disease progression, and 4 out of 5 patients with IIM reaching ACR major/moderate response. Furthermore, all patients remained free of glucocorticoids and any other immunosuppressive treatment over the entire observation period of 24 weeks. CASTLE suggests the feasibility, safety and efficacy of Zorpo-cel in three different autoimmune diseases and paves the way for conducting a pivotal study. ClinicalTrials.gov identifier: NCT06347718, EudraCT identifier: 2022-001366-35.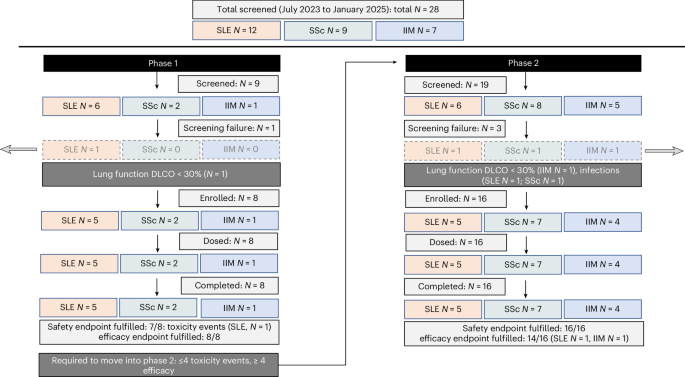
CD19 CAR-T cells for treatment-refractory autoimmune diseases: the phase 1/2 CASTLE basket trial - Nature Medicine
In the phase 1/2 CASTLE basket trial, autologous CD19 CAR-T cell therapy in patients with treatment-refractory systemic lupus erythematosus, systemic sclerosis or idiopathic inflammatory myopathy was safe, with improved disease activity and patient-reported global health in most patients.