Full title:
Evaluation of a Gene–Environment Interaction of PON1 and Low-Level Nerve Agent Exposure with Gulf War Illness: A Prevalence Case–Control Study Drawn from the U.S. Military Health Survey’s National Population Sample
https://ehp.niehs.nih.gov/doi/10.1289/EHP9009
Abstract
Background:
Consensus on the etiology of 1991 Gulf War illness (GWI) has been limited by lack of objective individual-level environmental exposure information and assumed recall bias.
Objectives:
We investigated a prestated hypothesis of the association of GWI with a gene–environment (GxE) interaction of the paraoxonase-1 (PON1) Q192R polymorphism and low-level nerve agent exposure.
Methods:
A prevalence sample of 508 GWI cases and 508 nonpaired controls was drawn from the 8,020 participants in the U.S. Military Health Survey, a representative sample survey of military veterans who served during the Gulf War. The PON1 Q192R genotype was measured by real-time polymerase chain reaction (RT-PCR), and the serum Q and R isoenzyme activity levels were measured with PON1-specific substrates. Low-level nerve agent exposure was estimated by survey questions on having heard nerve agent alarms during deployment.
Results:
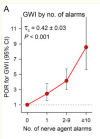
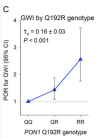
The GxE interaction of the Q192R genotype and hearing alarms was strongly associated with GWI on both the multiplicative [prevalence odds ratio (POR) of the interaction=3.41, 95% confidence interval (CI): 1.20, 9.72] and additive (synergy index=4.71, 95% CI: 1.82, 12.19) scales, adjusted for measured confounders. The Q192R genotype and the alarms variable were independent (adjusted POR in the controls=1.18, controls=1.18; 95% CI: 0.81, 1.73; p=0.35, and the associations of GWI with the number of R alleles and quartiles of Q isoenzyme were monotonic. The adjusted relative excess risk due to interaction (aRERI) was 7.69 (95% CI: 2.71, 19.13). Substituting Q isoenzyme activity for the genotype in the analyses corroborated the findings. Sensitivity analyses suggested that recall bias had forced the estimate of the GxE interaction toward the null and that unmeasured confounding is unlikely to account for the findings. We found a GxE interaction involving the Q-correlated PON1 diazoxonase activity and a weak possible GxE involving the Khamisiyah plume model, but none involving the PON1 R isoenzyme activity, arylesterase activity, paraoxonase activity, butyrylcholinesterase genotypes or enzyme activity, or pyridostigmine.
Discussion:
Given gene–environment independence and monotonicity, the unconfounded aRERI>0 supports a mechanistic interaction. Together with the direct evidence of exposure to fallout from bombing of chemical weapon storage facilities and the extensive toxicologic evidence of biochemical protection from organophosphates by the Q isoenzyme, the findings provide strong evidence for an etiologic role of low-level nerve agent in GWI. https://doi.org/10.1289/EHP9009
Full text on PMC
Evaluation of a Gene–Environment Interaction of PON1 and Low-Level Nerve Agent Exposure with Gulf War Illness: A Prevalence Case–Control Study Drawn from the U.S. Military Health Survey’s National Population Sample
https://ehp.niehs.nih.gov/doi/10.1289/EHP9009
Abstract
Background:
Consensus on the etiology of 1991 Gulf War illness (GWI) has been limited by lack of objective individual-level environmental exposure information and assumed recall bias.
Objectives:
We investigated a prestated hypothesis of the association of GWI with a gene–environment (GxE) interaction of the paraoxonase-1 (PON1) Q192R polymorphism and low-level nerve agent exposure.
Methods:
A prevalence sample of 508 GWI cases and 508 nonpaired controls was drawn from the 8,020 participants in the U.S. Military Health Survey, a representative sample survey of military veterans who served during the Gulf War. The PON1 Q192R genotype was measured by real-time polymerase chain reaction (RT-PCR), and the serum Q and R isoenzyme activity levels were measured with PON1-specific substrates. Low-level nerve agent exposure was estimated by survey questions on having heard nerve agent alarms during deployment.
Results:
The GxE interaction of the Q192R genotype and hearing alarms was strongly associated with GWI on both the multiplicative [prevalence odds ratio (POR) of the interaction=3.41, 95% confidence interval (CI): 1.20, 9.72] and additive (synergy index=4.71, 95% CI: 1.82, 12.19) scales, adjusted for measured confounders. The Q192R genotype and the alarms variable were independent (adjusted POR in the controls=1.18, controls=1.18; 95% CI: 0.81, 1.73; p=0.35, and the associations of GWI with the number of R alleles and quartiles of Q isoenzyme were monotonic. The adjusted relative excess risk due to interaction (aRERI) was 7.69 (95% CI: 2.71, 19.13). Substituting Q isoenzyme activity for the genotype in the analyses corroborated the findings. Sensitivity analyses suggested that recall bias had forced the estimate of the GxE interaction toward the null and that unmeasured confounding is unlikely to account for the findings. We found a GxE interaction involving the Q-correlated PON1 diazoxonase activity and a weak possible GxE involving the Khamisiyah plume model, but none involving the PON1 R isoenzyme activity, arylesterase activity, paraoxonase activity, butyrylcholinesterase genotypes or enzyme activity, or pyridostigmine.
Discussion:
Given gene–environment independence and monotonicity, the unconfounded aRERI>0 supports a mechanistic interaction. Together with the direct evidence of exposure to fallout from bombing of chemical weapon storage facilities and the extensive toxicologic evidence of biochemical protection from organophosphates by the Q isoenzyme, the findings provide strong evidence for an etiologic role of low-level nerve agent in GWI. https://doi.org/10.1289/EHP9009
Full text on PMC
Last edited by a moderator: