Nucleotide variations in genes encoding carbonic anhydrase 8 and 10 associated with femoral bone mineral density in Japanese female with osteoporosis
Abstract
Osteoporosis is a multi-factorial common disease, which is caused by combination of genetic as well as environmental factors. Among several factors, osteoclast acidification pathways during bone resorption might play an important role. Carbonic anhydrases, consisting of a gene family, are essential for pH regulation by the osteoclast. Clinically, use of carbonic anhydrase inhibitors has been known to be associated with a bone-sparing effect as judged by spine bone mineral density (BMD).
Here, we investigated single nucleotide polymorphisms (SNPs) in carbonic anhydrase genes that are expressed in bone tissues, i.e., CA8 and CA10, for possible association with femoral and lumbar BMD among 337 Japanese women with osteoporosis participated in BioBank Japan project.
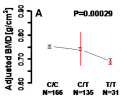
Significant correlation was observed between CA8 SNP, rs6984526, and femoral BMD (P = 0.00029); homozygous carriers of the major (C) allele (n = 166) had the highest BMD (0.754 ± 0.006 g/cm2, mean ± SD), while heterozygous carriers (n = 135) were intermediate (0.741 ± 0.07 g/cm2) and homozygous T-allele carriers (n = 31) had the lowest BMD (0.691 ± 0.012 g/cm2). CA8 SNP as well displayed significant association with lumbar BMD in recessive model (P = 0.00017). In addition, CA10 SNP, rs2106329, also displayed strong association with femoral BMD (P = 0.00002).
The results suggest that the variations of CA8 and CA10 loci may be important determinants of osteoporosis in Japanese women.
Web | DOI | PDF | Journal of Bone and Mineral Metabolism | Paywall
Mori, Seijiro; Kou, Ikuyo; Sato, Hidenori; Emi, Mitsuru; Ito, Hideki; Hosoi, Takayuki; Ikegawa, Shiro
Abstract
Osteoporosis is a multi-factorial common disease, which is caused by combination of genetic as well as environmental factors. Among several factors, osteoclast acidification pathways during bone resorption might play an important role. Carbonic anhydrases, consisting of a gene family, are essential for pH regulation by the osteoclast. Clinically, use of carbonic anhydrase inhibitors has been known to be associated with a bone-sparing effect as judged by spine bone mineral density (BMD).
Here, we investigated single nucleotide polymorphisms (SNPs) in carbonic anhydrase genes that are expressed in bone tissues, i.e., CA8 and CA10, for possible association with femoral and lumbar BMD among 337 Japanese women with osteoporosis participated in BioBank Japan project.
Significant correlation was observed between CA8 SNP, rs6984526, and femoral BMD (P = 0.00029); homozygous carriers of the major (C) allele (n = 166) had the highest BMD (0.754 ± 0.006 g/cm2, mean ± SD), while heterozygous carriers (n = 135) were intermediate (0.741 ± 0.07 g/cm2) and homozygous T-allele carriers (n = 31) had the lowest BMD (0.691 ± 0.012 g/cm2). CA8 SNP as well displayed significant association with lumbar BMD in recessive model (P = 0.00017). In addition, CA10 SNP, rs2106329, also displayed strong association with femoral BMD (P = 0.00002).
The results suggest that the variations of CA8 and CA10 loci may be important determinants of osteoporosis in Japanese women.
Web | DOI | PDF | Journal of Bone and Mineral Metabolism | Paywall