TiredMathematician
Established Member (Voting Rights)
Systematic Examination of Gene Expression and Proteomic Evidence Across Tissues Supports the Role of Mitochondrial Dysregulation in ME/CFS
Abstract
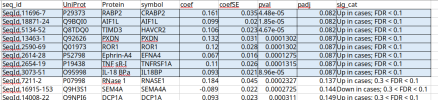
Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) is a chronic, multisystem disease characterized by post-exertional malaise and persistent fatigue. The cause of ME/CFS is not well understood, and there are no established biomarkers or FDA-approved pharmacotherapies. The clinical heterogeneity of ME/CFS presents challenges to diagnosis and treatment and necessitates collaborative efforts to generate robust findings. This study leveraged gene and protein expression data from the mapMECFS data repository and the DecodeME Genome-Wide Association Study (GWAS) to assess consistent gene signatures across studies. The mitochondrial genes MT-RNR1 and MT-RNR2 exhibited lower expression in ME/CFS cases in two studies. Combining this with increased expression of mitochondrial genes in platelets from another study, this supports mitochondrial dysregulation as having a role in ME/CFS. Furthermore, ME/CFS-associated genes were mapped to compounds in drug databases as possible treatments for further investigation. In muscle gene expression data, 107 approved compounds target 26 genes with functions relevant to mitochondrial support and immunomodulators. From the DecodeME GWAS, 83 approved compounds target 24 genes with functions related to energy metabolism and mitochondrial function. Though little consistency in specific genes was observed across studies, which highlights the need for larger studies, mitochondrial dysfunction in ME/CFS cases was evident across studies.
Web | DOI | International Journal of Molecular Sciences
Keele, Gregory R.; Enger, Mike; Barnette, Quinn; Ruiz-Esparza, Roman; Alvarado, Manuel; Mathur, Ravi; Stratford, Jeran K.; Giamberardino, Stephanie N.; Brown, Linda Morris; Webb, Bradley T.; Carnes, Megan Ulmer
Abstract
Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) is a chronic, multisystem disease characterized by post-exertional malaise and persistent fatigue. The cause of ME/CFS is not well understood, and there are no established biomarkers or FDA-approved pharmacotherapies. The clinical heterogeneity of ME/CFS presents challenges to diagnosis and treatment and necessitates collaborative efforts to generate robust findings. This study leveraged gene and protein expression data from the mapMECFS data repository and the DecodeME Genome-Wide Association Study (GWAS) to assess consistent gene signatures across studies. The mitochondrial genes MT-RNR1 and MT-RNR2 exhibited lower expression in ME/CFS cases in two studies. Combining this with increased expression of mitochondrial genes in platelets from another study, this supports mitochondrial dysregulation as having a role in ME/CFS. Furthermore, ME/CFS-associated genes were mapped to compounds in drug databases as possible treatments for further investigation. In muscle gene expression data, 107 approved compounds target 26 genes with functions relevant to mitochondrial support and immunomodulators. From the DecodeME GWAS, 83 approved compounds target 24 genes with functions related to energy metabolism and mitochondrial function. Though little consistency in specific genes was observed across studies, which highlights the need for larger studies, mitochondrial dysfunction in ME/CFS cases was evident across studies.
Web | DOI | International Journal of Molecular Sciences