ryanc97
Senior Member (Voting Rights)
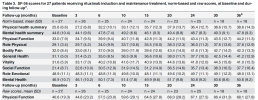
We have seen in 2022 Fluge and Mella wisely did a step count study with no intervnetion to understand the properties of natural step count variation in ME/CFS patients. And it looks like the average natural variation is around a 1k increase.
We have also seen in a placebo controlled study, improvements were on average a 1k increase too.
Now we have a small study with a 4k increase in average with 6 people. And people here are saying that this could still be due to chance.
Anyone up to buy some lottery tickets?
We have also seen in a placebo controlled study, improvements were on average a 1k increase too.
Now we have a small study with a 4k increase in average with 6 people. And people here are saying that this could still be due to chance.
Anyone up to buy some lottery tickets?