Trying to work out the make up of the ME/CFS group (I)
OK, I have done my homework
@Simon M! My brain is not working very well, so I have not done it as well as I would like, but hopefully it will still contribute to the conversation.
I agree with you that it is worth exploring the higher prevalence of ME/CFS at 6 months following infection reported in Jason 2020 compared to Katz 2009, Hickie 2006 and Pedersen 2019. Jason 2020 themselves write:
Another difference between our study and previous reports [2–5] was the high rate (23%) of ME/CFS following IM; this may be related to very close surveillance in our confined population of college students or high levels of baseline fatigue seen in college students [33].
Putting aside their proposed explanations for now, I think you’re right that the questions used to establish substantial reduction in functioning would allow people who do not have ME/CFS to meet the threshold. However, I'm not convinced that there is a threshold that can be established from questions like that, that would distinguish people who are recovering from IM slowly from people who have been ME/CFS'd.
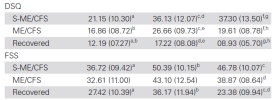
I think you’re right that the “(moderate) ME/CFS” group in Jason 2020 is likely composed of a mix of those with ME/CFS and other chronic fatigue, and that that may have artificially boosted the prevalence in Jason 2020. Your figures in
post 55 showing the baseline fatigue/DSQ etc of each group is particularly helpful here, though knowing what’s going on is a bit more complicated. Is there a premorbid subclinical difference between people who go on to develop ME/CFS and those who do not? Are some actually prodromal without knowing it? Or overexerting?
Having looked at all of the papers, I wonder if the problem starts further back, namely with the decision to give an ME/CFS diagnosis to people who fulfill
any of these criteria: Fukuda/IOM/Canadian. I’ll walk through my thinking on this.
Jason 2020’s methodology was most similar to that of Katz 2009 – unsurprising, as Katz was in both teams. Both studies had no initial exclusion criteria - meaning if you got IM, you were in, regardless of what other health issues you brought with you - a telephone screen at 5-6 months, and then a clinical examination of those not recovered.
In the telephone screen stage, in Jason 2020, 34.0% (81/238) were considered not fully recovered or had symptoms at 5 months vs 24.5% (70/286) at 6 months in Katz 2009.
In Jason 2020,
32.1% (26/81) had symptoms but were not considered to have Fukuda, IOM or Canadian-ME/CFS with substantial reduction in functioning, vs
44.3% (31/70) with symptoms but not considered to have Jason’s revision of Fukuda-CFS in Katz 2009. One possibility is that it was easier to get a diagnosis in Jason 2020, because you could get it with IOM criteria alone.
Edited to add this bit which somehow hadn't copied over:
In Jason 2020, we get no breakdown of how many met each set of criteria. All we know is this:
Note that in most cases the ME/CFS group only met the Fukuda criteria [20], while the S-ME/CFS group always met the Fukuda criteria [20] and either the Canadian [21] or IOM [22] criteria.
“Most” could mean anything from 51%-99%. So clarification from the authors on how many cases were diagnosed by IOM criteria alone (and Canadian criteria alone) would be welcome, as it would outrule one possible reason for the higher prevalence in Jason 2020.
The IOM criteria have been demonstrated, by Jason’s team (
Jason 2015), to be met by hefty proportions with other disorders:
33% with MS, 47% with lupus, 27% with major depression, 44% of those with idiopathic chronic fatigue not meeting Fukuda criteria and 47% with chronic fatigue due to other disorders.
The last two figures there are the most relevant for this case. You can see from this table that chronic fatigue that does not meet Fukuda criteria is extremely common:

Now, you can argue that anyone with those disorders would be weeded out by the physician assessment at 6 months. But establishing many of those alternative diagnoses would require a lot of consulations, and they might be in the early undiagnosable stages in college students. By contrast, Pedersen 2019 excluded 125 people with IM right off the bat, before they even entered the pool for the study:
Exclusion criteria were a) more than 6 weeks since debut of symptoms suggesting acute EBV infection; b) Any chronic disease that needed regular use of medication; c) Pregnancy.
In the Jason 2015 study just discussed:
A team of four physicians and a psychiatrist were responsible for making a final diagnosis with two physicians independently rating each file using the current U.S. case definition of CFS [
2]. Where physicians disagreed, a third physician rater was used [
3].
In Jason 2020, participants had “a comprehensive medical and psychiatric examination”, but we don’t get granular information on how many were diagnosed with other conditions, or how many fulfilled each set of criteria, or how many fulfilled certain criteria but were ruled out on the basis of not meeting reduced functioning thresholds, which would help us answer the questions you and I have raised.
Getting back to Katz, 12 of those 31 [from this figure
44.3% (31/70)] with symptoms but not considered to have Jason’s revision of Fukuda-CFS in Katz 2009 refused assessment, and one couldn’t do it in the right timeframe, so in theory, Katz’s prevalence could have been higher. For example, if 6 of them were diagnosed, then Katz’s prevalence would have been 15% (39+6)/301. [I see you covered this in a later post.]
What’s a bit tricky is that Katz 2009 accounts for each individual, but Jason 2020 does not. Here’s Katz et al.’s account:
Based on the screening interview, 70 of these adolescents (24%) were assessed as not fully recovered. A clinical evaluation was completed on 53 (76%) of these 70 not fully recovered adolescents; 12 refused, 3 had exclusionary diagnoses (primary depression, transverse myelitis, anorexia) and 2 did not meet study criteria (the fatigue predated the IM or the subject was not able to complete the 6 month evaluation in a timely fashion)…
Following the 6 month clinical evaluation, 39 of the 53 not fully recovered subjects who underwent clinical evaluation were classified as having CFS (13% of the original sample of 301 adolescents)…
Among the 14 other subjects completing the 6 month clinical evaluation, 1 had recovered between the time of the phone interview and the time he/she was seen in Clinic and 13 were classified as CFS explained (1 abused drugs, on more careful questioning 1 subject’s fatigue predated the mononucleosis, 1 had an eating disorder, 1 had an unrelated medical illness, 6 had underlying psychiatric diagnoses, 2 had psychiatric and sleep disorders, and 1 had an intercurrent acute parvovirus infection following IM, so symptoms could not be solely explained by infectious mononucleosis).
In Jason 2020 we don’t get that breakdown.
So I think it’s possible that some relatively small differences in how many alternative explanations were identified and considered exclusionary, and the ease of fulfilling IOM criteria with those unidentified alternative explanations, could contribute to a larger group being given the label ME/CFS than is warranted.
I'll have to come back to the explanations Jason et al. propose when brain working better.