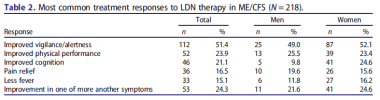
About half of the patients experienced a response with respect to at least two different ME/CFS symptoms. In responders, the extent of the response averaged 2.06 symptoms (men 2.19, women 2.03 symptoms).
18.3% of the sample did not report any response to LDN. 13.8% discontinued LDN for lack of effectiveness after an average of 5.9 months of use.
4.6% were unable to gain benefit because adverse symptoms forced them to discontinue the treatment during the introductory phase before the treatment response could be assessed (after an average of 1 month [1–57 days] of use).
Nearly half of the group (45.8%) did not experience any adverse symptoms from LDN treatment. The other 54.2% reported adverse symptoms, which weremostly mild and temporary. The adverse symptoms occurred only at the beginning of the therapy, or after dose increases. Typically, adverse symptoms were present for only a few days or a few weeks. In the worst cases, the adverse symptoms did not disappear until 1–2 months on treatment. Only one patient reported adverse symptoms during long-term treatment but preferred to stay on LDN in spite of mild nausea.
Serious adverse effects (SAEs) were not reported, although 7.3% of the patients discontinued treatment because of adverse symptoms. The most common reason for discontinuation was nausea (5 patients, 2.5%). Increased anxiety was the reason for early termination in two patients (1.0%).